|
5/31/2023 0 Comments Niels bohr atomic model The change in the energy of electron makes to jump from one orbit to another accompanied by either by radiation and absorption of energy.An electron’s angular momentum of an orbit is quantized and.An electron moves around the nucleus in a circular orbit.Bohr’s model of hydrogen atom is based on three postulates (Lee. Bohr’s theory explains the emission and absorption spectra of atomic hydrogen and hydrogen-like ions with low atomic number. He came to the conclusion that electrons will have more energy if they are placed away from the nucleus, while electrons will have less energy if they are located near the nucleus.īohr’s model of the hydrogen atom was the very first model of atomic structure that explained the spectrum of hydrogen model. He went over electrons and the various energy levels linked with them. As a result, Rutherford essentially explained an atom’s nucleus, whereas Bohr advanced the model. He also stated that each orbital shell has a set of energy levels. Rutherford’s concept, in which electrons travel around in fixed orbital shells, was amended by Bohr. The electrons follow a predetermined course known as orbits. Rutherford explained that in an atom, the nucleus is positively charged and surrounded by electrons in his Rutherford Model (negatively charged particles). Thomson’s discovery of the electron and Ernest Rutherford’s discovery of the nucleus. 
Many models of the atom have been proposed based on experimental data, including J.J. 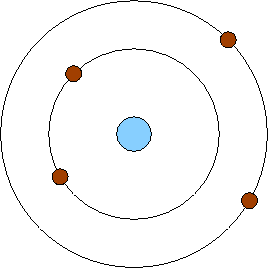 
Niels Bohr, who was interested in explaining the spectrum of discrete lines that is observed when light is emitted by different elements. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
